|
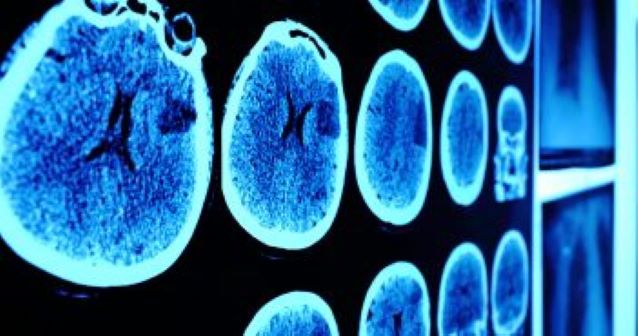
February 2021 STUDY IDENTIFIES NEW POTENTIAL THERAPEUTIC APPROACH FOR GLIOBLASTOMAFeaturing: Leonidas Platanias, MD, PhD and Frank Eckerdt, PhD
A team of Northwestern Medicine investigators have identified a potential combined targeted therapy approach for treating glioblastoma, the most common and lethal type of brain cancer in adults, according to a recent study published in the journal Scientific Reports. “There is an urgent need for innovative new approaches for the treatment of glioblastoma and our study raises the possibility of a new pharmacological combination approach. We hope that we will be able to eventually bring it to clinical trials to test its efficacy,” said senior author Leonidas Platanias, MD, PhD, the Jesse, Sara, Andrew, Abigail, Benjamin and Elizabeth Lurie Professor of Oncology, and director of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University. Platanias and study co-author Stewart Goldman, MD, chief of Hematology, Oncology, and Stem Cell Transplantation in the Department of Pediatrics, are also part of the Lou and Jean Malnati Brain Tumor Institute of the Lurie Cancer Center. There is currently no cure for glioblastoma and average life expectancy after diagnosis is 14 to 15 months. Over the last decade, however, scientists have made considerable progress in the characterization of glioblastoma on the molecular level and are now able to classify glioblastoma tumors into three major subtypes: proneural, mesenchymal and classical. Each tumor subtype is defined by different characteristics, including gene expression and the activation of various cell signaling pathways, which may aid scientists in determining how each subtype responds to specific therapies. Disease progression for glioblastoma is driven by glioma stem cells, a subpopulation of glioblastoma tumor cells that are notorious for being able to evade many treatment approaches. Glioma stem cells are not static but rather plastic, meaning they can evolve their characteristics and adapt to radiation and chemotherapy treatments. Due to their high plasticity, combined with their ability to constantly self-renew, glioma stem cells can give rise to “offspring” cells with even more therapeutic resistance and contribute to the disease’s overall treatment failure and cancer recurrence. “If you picture our fight against cancer as a competition of cleverness between cancer and medical professionals, then we are now trying to target the smartest subpopulation of cancer cells,” said lead author Frank Eckerdt, PhD, research assistant professor of Neurological Surgery, who is also a member of the Lurie Cancer Center. For the current study, investigators grew three-dimensional glioblastoma “neurospheres,” or glioma stem cell models, to identify any increased activation of specific cell signaling pathways and determine if targeting these pathways would prove effective in decreasing tumor cell growth. Using an advanced stem cell assay analysis technique, the investigators found increased activation of the PI3K/AKT signaling pathway — essential for regulating cancer cell division — specifically in glioma stem cells representing the proneural glioblastoma tumor subtype. According to Eckerdt, previous work has suggested that this same pathway is also an effective therapeutic target for treating medulloblastoma, the most common type of brain cancer in children. The team also found increased expression of the alpha catalytic PI3K isoform in proneural glioblastoma patient samples, which was associated with poor prognosis in this group of patients. To ascertain whether targeting this specific isoform might be a promising anti-cancer strategy specifically for proneural glioblastoma, the investigators used alpelisib, a drug currently used to treat certain types of breast cancer. Additionally, this drug selectively inhibits the alpha catalytic PI3K isoform and exhibits greatly reduced toxicity as compared to other PI3K inhibitors. The investigators found that alpelisib dramatically reduced growth of glioma stem cells of the proneural subtype. Futhermore, they discovered that the effects of alpelisib were enhanced in other glioblastoma subtypes when combined with pharmacological inhibition of mTOR, an enzyme that is essential for proper activation of additional cancer promoting cell signaling pathways. Overall, the current findings could serve as the foundation for future clinical trials, according to the authors. “Insights from studying these highly plastic glioma stem cells and their escape mechanisms may become an important part of our armory used to combating this deadly disease,” Eckerdt said. “Going forward, we plan to work closely with our colleagues in the Northwestern Brain Tumor SPORE to develop clinical translational applications relevant to these findings,” Platanias added. Co-authors of the study include Mariafausta Fischietti, PhD, research assistant professor of Medicine in the Division of Hematology and Oncology, and Goldman, who is also a member of the Lurie Cancer Center. This work was supported by grants from the National Cancer Institute and the National Institute for Neurological Disorders and Stroke. This article was originally published in the Feinberg School of Medicine News Center on February 4, 2021. |
Leonidas Platanias, MD, PhD, the Jesse, Sara, Andrew, Abigail, Benjamin and Elizabeth Lurie Professor of Oncology and director of the Robert H. Lurie Comprehensive Cancer Center, was senior author of the study published in Scientific Reports.
Frank Eckerdt, PhD, research assistant professor of Neurological Surgery and a member of the Robert H. Lurie Comprehensive Cancer Center, was lead author of the study.
Refer a PatientNorthwestern Medicine welcomes the opportunity to partner with you in caring for your patients.
|
You May Also Like
|
December 2020 |
December 2020 |