|
September 2020 NEW BIOMARKERS FOR GLIOMA TREATMENT RESPONSEFeaturing: Stewart Goldman, MD
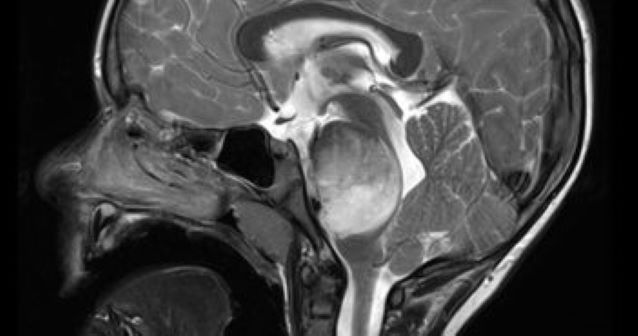
Biomarkers using mass cytometry can assess patient response to an emerging vaccine for a specific pediatric brain tumor, according to a recent multi-center study published in the Journal of Clinical Investigation. These findings show the vaccine is safe and the biomarkers could improve clinical trials for the peptide vaccine, according to Stewart Goldman, MD, chief of Hematology, Oncology, and Stem Cell Transplantation in the Department of Pediatrics and a co-author of the study. “These findings will allow future trials to better understand whom to offer these therapies to, and what medications we need to avoid as to not decrease efficacy,” said Goldman, who is also a member of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University. “ Diffuse midline gliomas (DMG) and diffuse intrinsic pontine gliomas (DIPG) are deadly pediatric brain cancers: less than 10 percent of patients survive beyond two years. Traditionally these tumors are treated with radiation therapy, but immunotherapy treatments have gained attention in recent years, according to the study. Previously, the study authors created a peptide vaccine for diffuse gliomas. This vaccine uses a mutation common in diffuse gliomas to trigger an immune response targeted at cancer cells — in essence, pointing the immune system at cells with this mutation and letting them attack the cancerous cells. In the current study, investigators tested safety and monitoring of the vaccine, enrolling 29 patients. The vaccine was well-tolerated with no serious adverse events, according to the study. In addition, immunological responses were measured using mass cytometry on blood cells. Mass cytometry allowed the investigators to measure how many T-cells were reactive to the cancer, as intended by the peptide vaccine. Increasing levels of reactive T-cells were associated with a better prognosis for patients, according to the study. Other cytometry measures included measuring myeloid cells to predict patient response to the treatment — demonstrating that mass cytometry on blood samples is a viable surveillance method, according to Goldman. “We showed that immunologic monitoring of patients receiving the vaccine therapy can reveal predictive biomarkers for response and efficacy,” Goldman said. Now that the vaccine has passed its first test, Goldman said he hopes it can move through the trial process and that other diffuse glioma treatments can take advantage of the monitoring framework laid out here. “This study clearly warrants further investigation and I hope using cytometry and other methodologies to monitor response will guide physicians in their therapeutic choices in the future,” Goldman said. This study was supported by the V Foundation, the Pacific Pediatric Neuro-Oncology Consortium Foundation, the Pediatric Brain Tumor Foundation, Mithil Prasad Foundation, the MCJ Amelior Foundation, the Anne and Jason Farber Foundation, Will Power Research Fund Inc., the Isabella Kerr Molina Foundation, the Parker Institute for Cancer Immunotherapy and the National Institutes of Health National Institute of Neurological Disorders and Stroke grant R35 NS105068. This article was originally published in the Feinberg School of Medicine News Center on September 15, 2020. |
Stewart Goldman, MD, chief of Hematology, Oncology, and Stem Cell Transplantation in the Department of Pediatrics, was a co-author of the study published in the Journal of Clinical Investigation.
Refer a PatientNorthwestern Medicine welcomes the opportunity to partner with you in caring for your patients.
|
You May Also Like
|
January 2021 |
February 2021 |